E-IRB News 2022
December 19, 2022
New Feature! All Attachments - Download Tool [HTML]
|
With the December 15, 2022 E-IRB system update, you can now download all attachments at once into a zip file. You can find the “Download all” tool in any location that lists all of the protocol’s attachments. Once you download and open the zip file, you can open all of the files or just selected items.
As protocol attachments are not included in the compiled ProtocolPDF file, this tool should be useful for anyone who doesn’t want to download application files one by one. |
November 29, 2022
New Feature! Ability to Import Study Personnel [HTML]
September 8, 2022
September 7th System Update [HTML]
|
Effective September 7, 2022, following the E-IRB system update, researchers will no longer receive the message “There is a problem uploading this attachment; e.g., password protected, portfolio PDFs, corrupt files, and some digitally signed files are not supported” when attempting to upload application attachments if the files fall into one of those restricted categories. Upload restrictions will only be applied to attachments a researcher is trying to upload to an Other Review (i.e., Protocol Violation (PV); Unanticipated Problem (UP); Deviation/Exception (DEV/EXC); or Administrative Study Closure). If you have created an Other Review and need to upload an attachment that falls in one of the restricted file categories, please either scan the document and upload the scanned version as a PDF file, or create a screen shot of the document and upload the screen shot as a PDF file. [EDIT: Files can also be printed to PDF and uploaded.] |
August 17, 2022
E-IRB Version 4 Update 8-18-22 [HTML]
|
IMPORTANT INFORMATION FOR SUBMISSIONS IN PROGRESS As previously announced, the Version 4 (V4) system update on August 18, 2022 encompasses improvements to the IRB application’s readability by reducing redundancies; re-organizing some of the sections; and improving navigation for some of the menu options, visuals on section pages, and the log-on page.
If one or more of these nine sections had a check mark before noon, August 18th, the section will lose the checkmark after the conversion to V4. A blue check mark indicates the section is complete while a pencil icon signifies that additional information/verification is required.  The researcher is responsible for going through the application to make sure all sections have a blue check mark next to them rather than a pencil. If any section in the application has a pencil icon next to it, it means there is missing information (e.g., a new question was added). The researcher will need to review the section and answer the required questions and save the section before being able to submit the application. As always, we appreciate your patience through the transition and hope you find the changes beneficial. |
August 2, 2022
E-IRB Version 4: Update Rescheduled [HTML]
|
An announcement was issued in early July about the implementation of a new E-IRB version “Version 4” (V4) tentatively scheduled for August 3rd. V4 encompasses improvements to the IRB application’s readability by reducing redundancies, re-organizing some of the sections, and improving the navigation for some of the menu options and the visuals on section pages. The upgrade to V4 has been rescheduled for August 18th to ensure appropriate implementation. The update will involve approximately three hours of offline time for the conversion and testing starting at noon on the 18th, so please adjust your schedule accordingly to accommodate the interruption in access to E-IRB. As the implementation date approaches, a system message will appear on your E-IRB Dashboard as a reminder. For a detailed recap on what V4 entails, you may wish to review the July 8, 2022 Online Edge newsletter. As always, we appreciate your patience as we work to get the planned improvements underway! |
July 8, 2022
E-IRB Version 4: Coming Soon [HTML]
|
Over the last several months, the Office of Research Integrity has focused on making improvements to the IRB application’s readability by reducing redundancies and re-organizing some of the sections. The tentative date for implementation is August 3, 2022. Putting this version (“V4”) of E-IRB into production will affect what you see and experience as follows:
At a minimum, the sections that will lose the check mark:
Additional changes in what you see in E-IRB include the log-in page and some of the menu selections on the researcher Dashboard. Another announcement will follow in the coming weeks with additional details and to confirm the implementation date. We look forward to getting the improvements underway! |
March 3, 2022
Research Reports [HTML]
E-IRB has a new report portal for ease of access to key Tableau Server reports.
On your Researcher Dashboard, you will notice a new menu option called “Tableau Reports”.
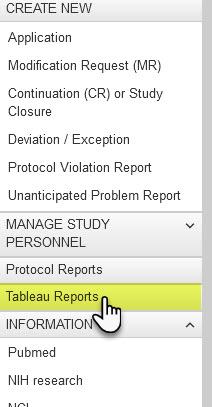
This link opens a page with a table including a link to Tableau Server resources (e.g., “ABC’s” on how to gain access & use Tableau) followed by a list of reports available to you in your Researcher role. Reports accessed in your Researcher role will only return results for applications you have permission to access. More reports than what are listed below may be added in the future.
| Name of the Report | URL | Description of the Report |
|---|---|---|
| ABC's of Tableau Reports | Click here [HTML] | Review online resources on how to get a Tableau Server account in order to access data in the below reports, as well as use of Tableau Server. |
| Personnel Not Trained | Click here [HTML] | Review Personnel listed on my studies that are not current on the mandatory Human Subject Protections (HSP) training. |
| Dept Authorization - Applications for which I Signed an Assurance | Click here [HTML] | Look up applications for which I completed an Assurance Statement in a 'Department Authorization', 'Faculty Advisor', or 'Review by Other' role in the Signatures (Assurances) section of the E-IRB application. |
The Tableau Server reports aim to supplement “Protocol Reports” with search filters on various attributes in your application(s):
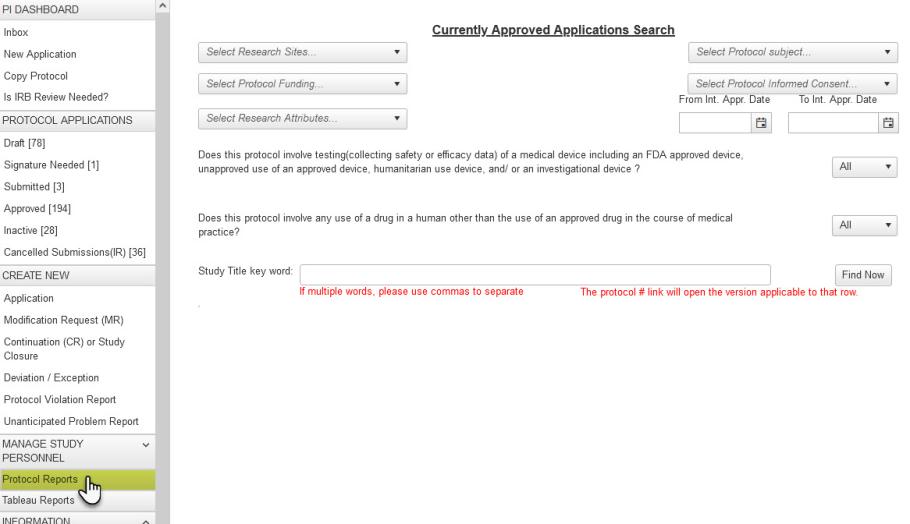
Questions about Tableau that you don’t find answered in Institutional Research, Analytics, and Decision Support (IRADS) resources should be directed to IRADS staff via their “Contact Us” tool. We hope the new Tableau Reports feature enhances your E-IRB experience!


