FAQs Initial CITI HSP Training for IRB Approval
How do I enroll in initial (basic) human subject training for IRB approval on the CITI website?
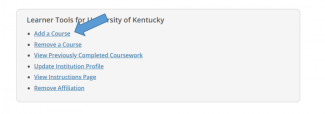
You may enroll initially from the Courses page after logging in to your account. Scroll to the bottom of your Courses page then click "Add a Course".
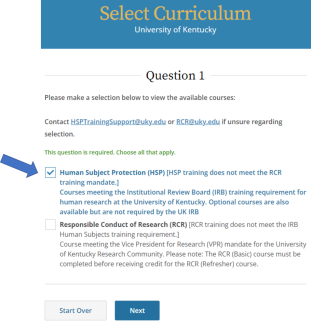
In the next window, select the “Human Subject Protection (HSP)” menu, then click Next.
NOTE: Do not select the “Responsible Conduct of Research (RCR)” menu, which offers RCR training options. RCR training does not meet the IRB Human Subject Protection training requirement.
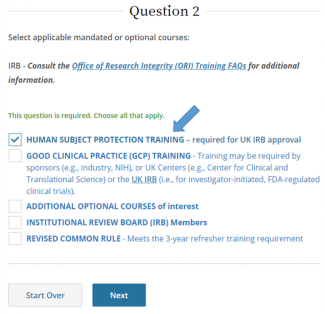
Select Human Subject Protection Training, then Next.
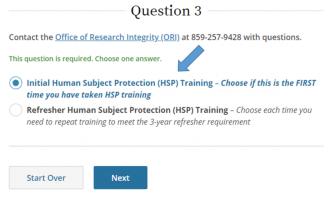
Select Initial Human Subject Protection Training, then Next.
Select the appropriate training for your research, then Next
You are directed back to your Courses page where it states you are now enrolled.
Scroll down to "Courses Ready to Begin" and click Start Now to complete the Initial HSP training.
The UK curriculum on CITI offers two course menus, Human Subject Protection (HSP) and Responsible Conduct of Research (RCR). Which do I choose to access HSP training for IRB approval?
To access HSP training required for IRB approval, select "Human Subject Protection (HSP)" and NOT "Responsible Conduct of Research (RCR)" from the CITI curriculum page. RCR training does NOT meet the training requirement for IRB approval. RCR stands for Responsible Conduct in Research. RCR training required in order to receive grant funding from select agencies.
I took the CITI Responsible Conduct in Research (RCR) course. Will that count for IRB approval?
No, if your intent is to complete IRB human subject protection training, be sure to select "Human Subject Protection (HSP)" from the CITI curriculum page.
Responsible Conduct in Research (RCR) training covers topics integral to the preparation and development of current and future scientists in all areas of research (not just human subjects). It is a requirement of certain funding agencies such as the National Science Foundation (NSF). For information on the RCR training or requirement, see the RCR question under the Other Training FAQs.
I took the CITI Good Clinical Practice (GCP) course. Will that count for IRB approval?
No. For IRB approval, you must complete human subject protection (HSP) training. GCP training is offered as an optional course that users may choose to take in addition to HSP training. GCP is an excellent training for those involved in the conduct of clinical research trials. Some industry sponsors of clinical trials require research staff to be trained in GCP guidelines. The CITI GCP course is one option for obtaining GCP training.
I took the CITI HIPAA course. Will that count for IRB approval?
No. For IRB approval, you must complete human subject protection (HSP) training. HIPAA training is offered as an optional course that users may choose to take in addition to HSP training. The CITI HIPAA training provides general information regarding the privacy rules and security rules.
Is there a paper-based alternative for initial human subject protection (HSP) training if I do not have access to a computer to complete CITI?
Study personnel are encouraged to complete the initial HSP training on CITI. If extenuating circumstances prohibit access to a computer, contact Belinda Smith to obtain an alternate paper-based course and exam.