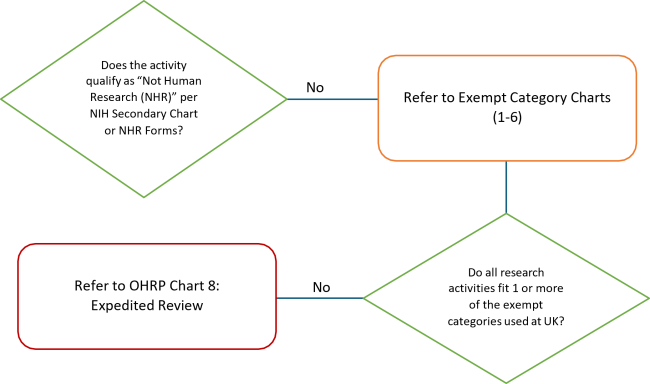
- Some activities that do not meet the regulatory definitions of research and/or human subjects may not require IRB review. See the "What Needs IRB Review" page and/or submit a Not Human Research (NHR) Determination Form for an official ruling on whether an activity is human subject research requiring IRB review.
- Researchers may not make a self-determination that their research qualifies for Exempt review. See the following Exempt Category Charts, refer to the ORI Exempt Review Decision Tool, and/or contact the UK Office of Research Integrity (ORI).
- In order for the research to be eligible for exemption, the only involvement of human participants in the research must fall into one or more of the exemption categories.
- Subpart B: Studies Involving Pregnant Women, Fetuses, & Neonates are Eligible for Exemption Under All 8 Categories
- Subpart C: Exemptions Do Not Apply to Research Involving Prisoners Except “for Research Aimed at Involving a Broader Subject Population that Only Incidentally Includes Prisoners.”
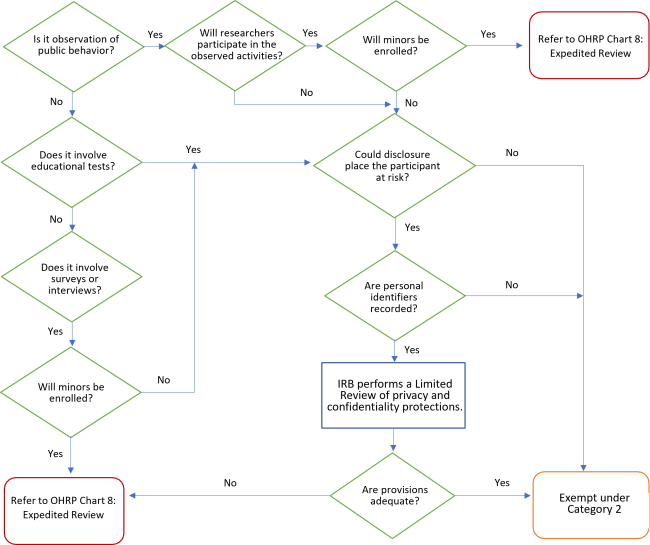
- Subpart D: Children are allowed in categories 1,4,5,6,7, & 8; Limitations & Exclusion of Children in Category 2 & 3
2020: UK ORI Exempt Decision Charts are based solely on the current interpretation of the revised Common Rule regulation (absent federal guidance or review). Select charts adapted with permission from Cornell University IRB. Contact Belinda Smith with questions.