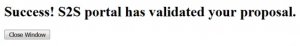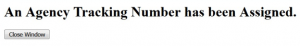A complete and final proposal, accompanied by a fully routed Internal Approval Form (IAF), must be received by the Research Administrator (RA) in OSPA by 9:00 am at least three (3) business days prior to the sponsor’s deadline. For more information about the 3-day Proposal deadline, click here.
Step 1: Prior to Submission
Keeping in mind the 3-Day Proposal Deadline, work with your Grant Proposal Specialist (GPS) to ensure that your proposal is eligible for PI submission. For more information about the 3-Day Proposal Deadline, click here.
- Proposals using the SF 424 R&R form are eligible for submission by PI.
- Proposals using the Application for Federal Assistance SF-424 form that meet the criteria below are not eligible for submission by PI:
- A separate field is not identified for uploading the budget justification.
- All application components are uploaded as one complete package.
- All application components are uploaded as separate documents utilizing the ‘Other Attachments’ form.
Step 2: Prepare your Proposal
Keeping in mind the 3-Day Proposal Deadline, work with your Grant Proposal Specialist (GPS) to prepare your proposal and ensure that all administrative fields are free of errors. For more information about the 3-Day Proposal Deadline, click here.
Step 3: Proposal Routing & Approval
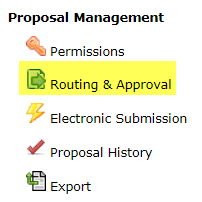
Once your proposal is free of errors, a “routing chain” will be established. The OSPA Research Administrator for your area must approve the proposal before you can submit. If routing hasn’t been created, then:
- Select the “Routing & Approval” link.
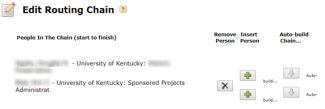
- Check the routing chain and edit as needed; it must always begin with the PI and end with your OSPA Research Administrator.
Step 4: Submitting
Important Note: Submission requires that the browser used accepts pop-ups from Cayuse. Please ensure that any pop-up blockers are set accordingly.
When ready to submit, do the following:
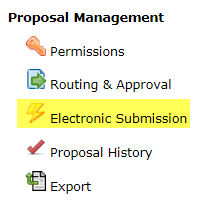
- On the left-hand side navigation pane, scroll down to Proposal Management and select the lightning bolt icon that says Electronic Submission. This will take you to the submission screen.
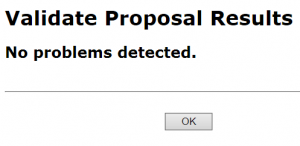
- In the Electronic Submission screen click the Validate Proposal button to check for errors one last time. You should get the message that there are no errors. If you do receive an error message you will need to correct the error before you can submit.
A pop-up screen will appear with validation results.
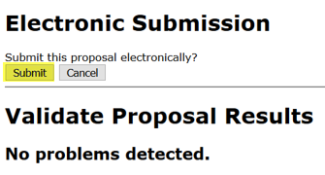
- Then check the box next to Submit to Grants.gov after Validation and click the Validate Proposal button again.
- A pop-up screen will appear for submission. Click Submit.
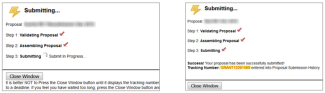
- The following submission steps will appear.
Wait until you get your tracking number before closing the screen. The tracking number will be in the format of GRANT0012345.
- Close the pop-up screen and click the + sign next to Proposal Submission History. You will see your tracking number as a blue link.
You can click on it to check the status of transmission, and you may want to do that intermittently to make sure that the application is validated.
- Validation message #1: Validation by Grants.gov
- Validation message #2: Agency Retrieval
The agency retrieval is generally the final message and means the proposal was successfully submitted.
For NIH submissions, an "Agency Tracking Number Assignment" email should be received. Be sure to check your proposal in eRA Commons.
Times for receipt of the messages and agency retrieval vary. Messages may take minutes or hours and occasionally part of a day.
Ready to submit? Log in to Cayuse at https://uky.cayuse424.com/